Dr. Stephany Veuger and Dr. Gina Abdelaal from Northumbria University describe their 2021 review published by Oncotarget, entitled, “Reversing oncogenic transformation with iron chelation”
Behind the Study is a series of transcribed videos from researchers elaborating on their oncology-focused studies published by Oncotarget. Visit the Oncotarget YouTube channel for more insights from outstanding authors.
—
Hi, my name is Dr. Stephany Veuger. I’m a Senior Lecturer at the University of Northumbria in Newcastle upon Tyne in the United Kingdom. Pretty soon I’ll introduce you to my PhD student, Gina Abdelaal, who is a student in my lab working with another student, and she’s the first author of the review published in 2020 which is entitled Reversing oncogenic transformation with iron chelation. I’ll introduce Gina pretty soon, who will talk to you about her research area. What she’s found, her key findings in the review, we hope to achieve in the lab in the future.
So my work, just to take a little bit of a step back and talk about where I’m coming from, supports the primary goal of anti-cancer drugs. And so by that, I mean to selectively interact with molecules essential for cancer cell survival. I have extensive background in cancer drug discovery. I have extensive experience in preclinical evaluation of novel therapeutics, this is with DNA repair inhibitors. And lots of my research has been looking at those drugs, as potential to be used with combination with current cancer therapies or as standalone therapies in their own right.
I’ve supported anti-cancer drug discovery projects in many different ways. For example, by investigating their interactive effects of proteins involved in sensing and responding to DNA damage, by developing and validating pharmacodynamic biomarkers, agents progressing to clinical trials, and also by evaluating enzyme inhibitors designed to exploit underlying molecular defects in a range of human cancers. But overall, the key focus of my research has always been proof of principal testing of preclinical lead compounds. This will lead into discussing both the review and the work that Gina is currently doing.
The work that I’ve done evaluating agents progressing to clinical trials and in helping to develop biomarkers has contributed towards, for example, the successful introduction of poly (ADP-ribose) polymerase. Or let’s call them PARP inhibitors, into patients. And most recently in 2019, rucaparib, which is a first-class PARP inhibitor that I was involved in the in vitro testing of, was announced by Clovis Oncology to be made available for women.
So the major research achievements that I’ve had are, first of all, as a young PhD student, was to show the identification of a competition as well as a cooperation between two particular DNA repair enzymes. So polymerase (ADP-ribose) that I’ve already introduced, and another enzyme called DNA-dependent protein kinase. And what this work did was further the understanding that we see when we have overlapping and competing pathways in cancer cell signaling. In particular, it highlighted the potential for combination therapies that can be used to treat cancer.
I was also involved in demonstrating for the first time that enzyme PARP-1 has a role in DNA double-strand break repair. And I have to admit this data was received with a lot of apprehension at first, amidst a huge amount of evidence that PARP was mainly only involved in base excision repair. This observation was recognized widely as needing further investigation, and many studies did actually swiftly build on that work that I had done. From there, this led to the important identification of a backup role for PARP in homologous recombination repair, and showing this homologous recombination repair-defective tumors were far more sensitive to PARP inhibitors than those that were not defective. And thus the role for standalone therapy with PARP inhibitors, particularly in these BRCA-deficient breast and ovarian tumors, was born.
One of the other things that I’ve been involved, demonstrating a very controversial role for PARP-1 and another enzyme. Ataxia telangiectasia-mutated, ATM, the way in which they mediate cell survival via nuclear factor kappa B, and not through the widely purported DNA repair pathways that everybody was publishing on. The reason I mention that is because although it was met with great skepticism at the time and it’s only recently become a field of study and an approach to mediate cancer therapy, why … the reason that work was so important was that it highlighted the need for a molecular understanding of aberrant singling pathways in cancer, in order for targeted that cancer therapy to work.
So, where am I now? Well, currently I have a number of collaborations with synthetic chemists, both at Northumbria University and at Newcastle University. And here at Northumbria University, we’re exploring the underlying biological mechanisms of novel therapeutics, both alone and in combination with some of those repair inhibitors that I’ve mentioned. And this portfolio of research that I have includes exploring the LAT1 transporter, as a novel mode of entry for ion chelators, to enable selective targeting of those tumors that actually overexpress this transporter.
The data that we’re going to be talking about today is being generated by three PhD students, two of whom are currently in the lab. And one of those students is Gina Abdelaal, who will come on and speak to you further about this in a moment. Gina and my other PhD student, Andrew Carter, are furthering the work of a chemistry PhD student who are trained up in some of the key techniques needed to accrue data quickly. So just to give a very brief overview of where we were at, at the point of the review being published before Gina takes over, our initial discovery of these hydroxypyridinone-based chelators which are LAT1 substrates, we were able to demonstrate their cytotoxic activity and selectivity for cancer cells over healthy cells in melanoma.
So, the original model was in melanoma. Ongoing preliminary work by myself and Gina has identified activity against a variety of other cancerous cell lines, including prostate cancer. And some of that preliminary data within our group is showing that a more aggressive PC3 cell line is more sensitive than a less aggressive DU145 cell line, so demonstrating that these compounds can have profound effects on cell survival and death. Being a little bit more specific then, we have published on melanoma. And shown that the lead compound we have, SK4, has the ability to one, promote increased generation of reactive oxygen species. Two, activate both intrinsic and extrinsic apoptosis.
And finally, they’re also able to induce perturbations in cell cycle and induce growth arrest. So what I’m going to do now is pause here and change over, across to Gina to talk a little bit more in detail.
I’m Gina Abdelaal, and I’m a third-year PhD student at Dr. Veuger’s lab at Northumbria University in the UK. I will give a brief overview of the review that we have published together and some of the back round behind it.
So iron chelators are a set of cancer drugs which strip cells of their iron content, with some also inducing reactive oxygen species. And the rationale behind their use is in cancer therapy, is cancer cells usually have a higher level of iron compared to normal cells. Usually because cancers upregulate their iron import proteins, such as transferrin, and downregulate their iron export proteins. And this accumulation of iron is what helps support cancer cell growth and metabolism, as many enzymes in the cells are iron independent.
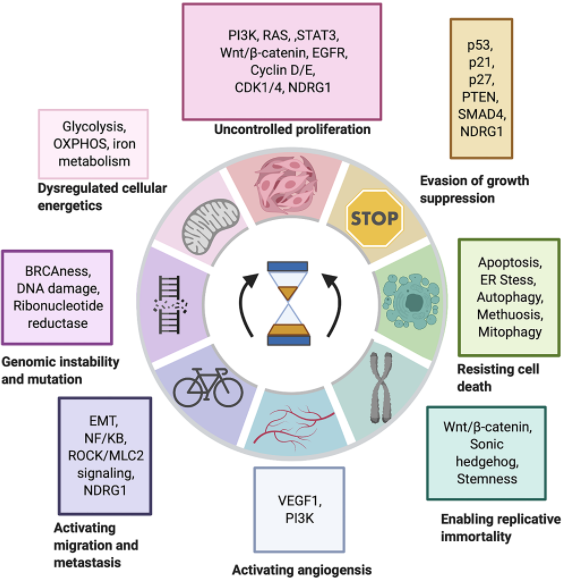
In DNA synthesis for example, is reliant on ribonucleotide reductase, which produces dNTPs. This enzyme uses iron as a cofactor, and this is the rate limiting step of DNA synthesis. Therefore iron can be a major driver of cell proliferation, and different tumor types may accumulate iron or be addicted to iron. So iron chelators are an effective agent against many tumor types, because it’s a universal trait. And that makes them a tumor-agnostic therapy, even with p53-mutant cancers. Our review entitled Reversing oncogenic transformation with iron chelation investigated the mechanistic side of iron chelators and related them to the hallmarks of cancer.
Iron chelators have been shown to reverse some of the hallmarks of cancer through a cell signaling protein called NDRG1, essentially when cells are treated with iron collators in vitro. And DRG1 is upregulated, through which many oncogenic signaling pathways are inhibited. Such as Wnt/β-catenin signaling, PI3K, and EGFR signaling. However NDRG1’s role as a tumor suppressor is still controversial, as some studies suggest more of a oncogenic sort of role. In some patient samples, you see an upregulation of NDRG1 being associated with poor prognosis. And for others, high NDRG1 means good prognosis. So, there’s still some controversy there.
In triple-negative breast cancer cells, iron chelation actually induced an upregulation of PI3K signaling and induced transferrin expression. This made those triple-negative breast cancer cells accumulate more iron, so there’s still room for debate on NDRG1’s exact role, and it may not be consistent across all cancer types. Additionally, iron chelators can induce apoptosis, autophagy and also ER stress. We’ve also seen that when you combine iron chelators with PARP inhibition, there is an increase in sensitivity in BRCA-positive cell lines which would normally be resistant to the PARP inhibitors. So, what I would really like to see next is more focus on clinical trials that use iron chelators in combination with chemotherapeutic drugs or possibly other anti-cancer agents.
That we’ve seen a mixed bag of results with clinical trials with iron chelators, there’s been a lack of response in some cases, as well as resistance and some side effects. Potentially, I would really like to see more focus on making iron chelators more selective. There are some studies that have shown if you encapsulate an iron chelator in nanoparticles, it makes them slightly more selective and effective against cancer cells. At the moment, I am working alongside another PhD student. Andrew Carter who’s, Dr. Veuger has mentioned. And I’m working on investigating the effects of SK4, which is based on alimemazine and has a moiety that allows entry through the amino acid transporter LAT1. SK4 is aimed at targeting LAT1-overexpressing tumors, making it selectively cytotoxic potentially in clinic.
Dr. Stephany Veuger
Okay. So I just wanted to really say thank you to everybody who’s been involved in this research with SK4 – which is an iron chelator with a LAT1 moiety attached to it – which we hope to be far more selective for cancer cells than perhaps some of the other iron chelators that are out there at the moment.
So I’d like to say thank you to David Tetard who is our synthetic chemist, who has designed, synthesized and worked really hard to produce enough compound for us to test. And then our previous PhD students, Sotiris Kyriakou and his supervisor, Mihalis Panayiotidis for all of their work underpinning a lot of the pilot data in melanoma that has enabled us to continue to look at this in more depth. In other cell lines, and then go on to look at the mechanisms. I wanted to thank all those that have been involved. Thank you.
TESTIMONIAL: Dr. Abdelaal Describes Publishing With Oncotarget
Click here to read the full research paper, published by Oncotarget.
ONCOTARGET VIDEOS: YouTube | LabTube | Oncotarget.com
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues, and other researchers, far and wide.
For media inquiries, please contact media@impactjournals.com.