Behind the Study is a series of transcribed videos from researchers elaborating on their recent oncology-focused studies published by Oncotarget. A new Behind the Study is released each Monday. Visit the Oncotarget YouTube channel for more insights from outstanding authors.
—
Dr. Aditya Kulkarni, Senior Research Scientist at Lantern Pharma, discusses a study he co-authored, published by Oncotarget in 2021, and entitled, “The acylfulvene alkylating agent, LP-184, retains nanomolar potency in non-small cell lung cancer carrying otherwise therapy-refractory mutations.”
Hello everybody. My name is Aditya Kulkarni and I’m a Senior Research Scientist at Lantern Pharma. I’d like to thank the Oncotarget journal and its associated publishing house for the opportunity to present our cancer drug development research activities on this platform.
I’ve been a biopharmaceutical scientist, manager and inventor combining over 12 years of research experience with a vision to direct drug candidates from the bench to the bedside. I am a biochemist by training, having applied it in the fields of cancer biology and small molecule drug development. One of my areas of interest involve identification and validation of biomarkers of drug response and resistance and at Lantern Pharma I’ve been fortunate to have explored and contributed to several areas of drug development. Lantern Pharma is a clinical stage oncology-focused biopharma company, leveraging artificial intelligence methodologies to generate gene signatures, predictive of response in patients to its pipeline of genomically-targeted therapeutics.
Using this precision medicine approach, Lantern Pharma aims to potentially deliver best in class outcomes using the method of targeting drugs to patients whose genomic profile is going to identify them as having the highest probability of benefiting from a given drug. Lantern Pharma is having four drug candidates in development, all in various cancer types, having unique and unmet clinical needs. One of these is LP-184, which has a very interesting history as well as a unique anticancer activity profile, which brings me to the topic of our publication.
The title of the paper, which I’m going to be describing in today’s session, is “LP184 retains nanomolar potency in non-small cell lung cancer caring otherwise therapy refractory mutations.” And as the lead author on this study, I’ll make sure to break down the title for everybody to appreciate. Our paper is essentially describing the activity of LP-184 Lantern Pharma’s lead pre-clinical drug candidate in a wide range of preclinical models of lung cancer, specifically non-small cell lung cancer, which accounts for 85 to 90% of the total lung cancer incidents. LP-184 belongs to a class of compounds called acylfulvene agents, which further can be categorized as DNA damaging agents.
In this paper, we also identify genetic correlates and determinants of LP-184 sensitivity that will facilitate further biomarker development during clinical translation and allow additional personalization of therapeutic options for future treatment of non-small cell lung cancer. With this, I’d like to talk about a bit of a history of the research leading up to this paper.
Our company, named Lantern Pharma, actually comes from the lantern mushroom that naturally produces a fungal toxin called illuden. The first identified examples of illudens were isolated the lantern mushroom at the New York Botanical Garden in the 1950s. Illudens were then extensively studied for their cytotoxicity in various tumor types, in cell lines, as well as xenograft models.
However, frequent animal deaths associated with high illuden toxicity restricted their use as potential anticancer agents. In an effort to obtain cytotoxic compounds with favorable anticancer properties, acylfulvenes were derived as semi-synthetic analogs from illudens and this class of compounds with the core structure of acylfulvenes and their analogs were much milder cytotoxins than their natural precursors, illudens and exhibited favorable tumor specificities.
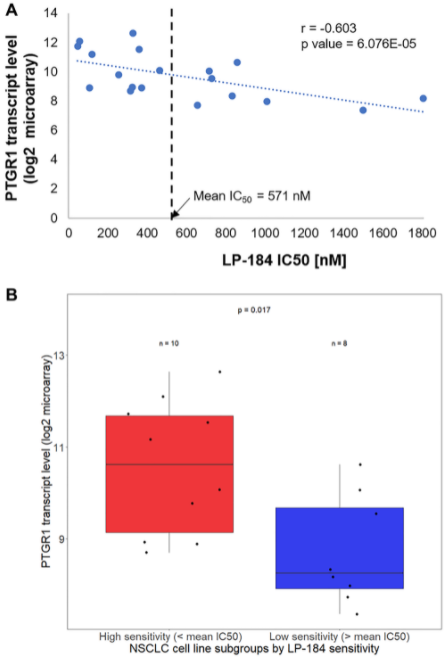
Now I want to point out four highlights that emerged from this early generation of acylfulvene molecules: Number one is the retention of sensitivity of acylfulvene molecules in multi-drug resistant cancer cells that were otherwise resistant to commonly used chemotherapy agents, such as cisplatin, doxorubicin, and irinotecan. The second highlight is that as acylfulvenes are uniquely tumor-targeting while sparing normal or non-tumor cells. The third highlight is that as acylfulvenes show heightened activity in cancer cells with compromised DNA repair capacities. And the fourth highlight, which is the most remarkable, is that acylfulvenes are pro-drugs that are activated by a prostate gland in reductase-1 or PTGR1, which is an oxidoreductase enzyme and has now been established as a fundamental driver of tumor sensitivity and appears to be a unique and stringent efficacy biomarker for this class of compounds.
High level of interest in this profile of acylfulvene bioactivity and more of action further triggered the development of new synthetic strategies to produce newer generations of acylfulvenes and their analog. We identified LP-184 as a next generation acylfulvene analog with highly improved and favorable therapeutic window potency, and a validated role of PTGR1 in its cytotoxicity, along with preliminary hints of synthetic lethal relationship with DNA damage repair defects. It isn’t surprising that certain mutations in oncogenes and tumor suppressor genes underlie non-small cell lung cancer, likely by acting as drivers of tumorigenesis, or metastasis, or therapy resistance at some point in the disease and treatment progression. But what is surprising is that if we consider just four genes; two oncogenes, KRAS and keep one, and two tumor suppressor genes; TP53 and STK11, these combined account for more than 40% of non-small cell lung cancer cases.
And it is not surprising that alterations in tumor suppressors or oncogenes underline non-small cell lung cancer, likely by driving tumorigenesis, metastases, or treatment resistance at some point during the disease and treatment progression. What is surprising is that if you consider a handful of these tumor suppressor or oncogenes, namely KP1 and KRAS that are famous oncogenes and TP53 and STK11 that are tumor suppressors, these four genes account for more than 40% of non-small cell lung cancer cases. And there are hardly any therapies that are effective in patients having mutations in these genes. Furthermore, it’s important to note that the signaling pathways and interaction networks in which these genes operate are non-overlapping or non-redundant, and hence, it’s very difficult to develop any single class of agents to target non-small cell lung cancer having alterations in these genes. And we believe that LP-184 could really have a big impact, potentially in these molecular subsets that appear to be very segregated.
I wish to talk about the most notable features of our work published in this paper and that can be summarized in six points.
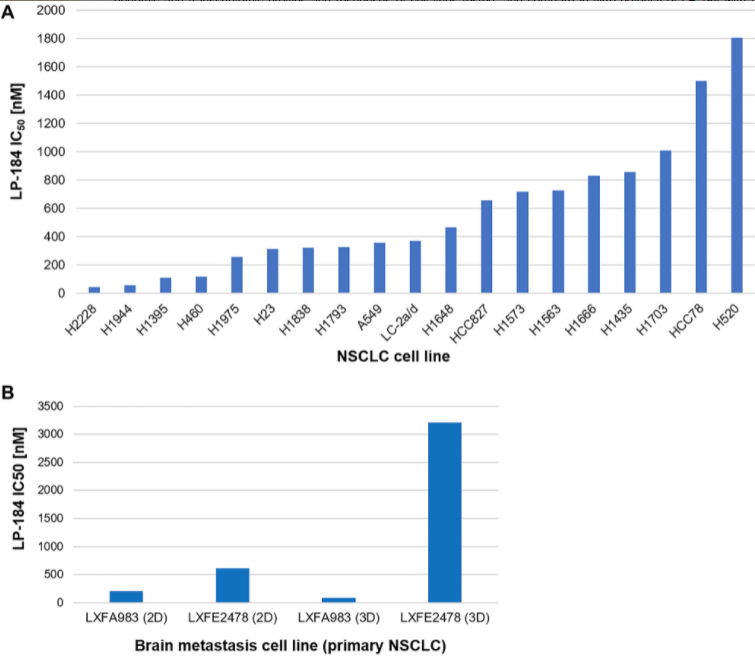
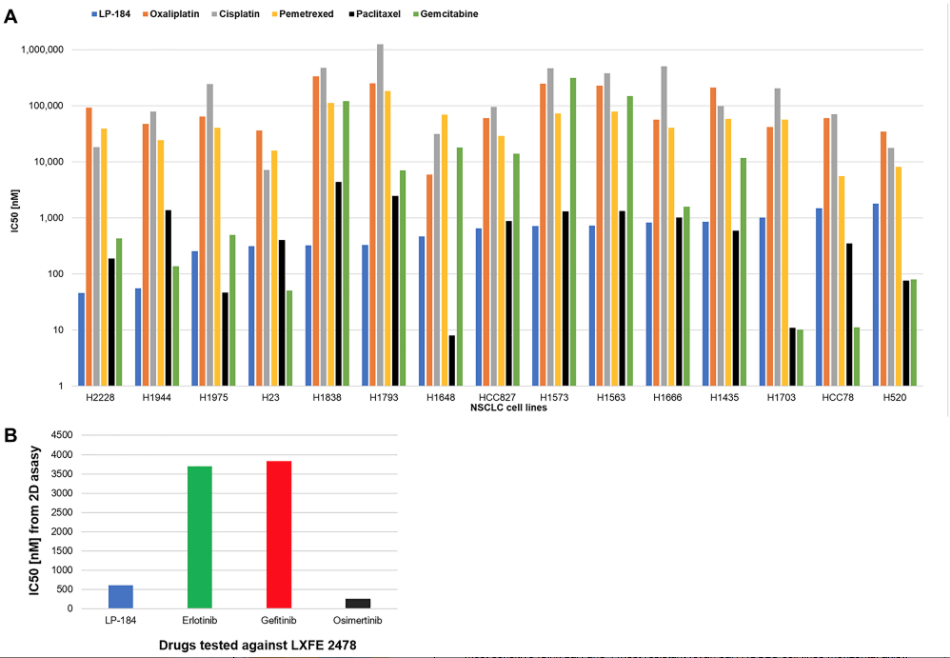
One is that LP-184 is effective in vitro, not only in 2-D models, and in primary non-small cell lung cancer models, but also in 3-D models of metastatic lung cancer models, especially brain metastasis originating from primary non-small cell lung cancer. And this is especially relevant for application in brain metastasis since we also found that LP-184 crosses the blood-brain barrier in an in vitro model of multicellular human blood-brain barrier. The second feature of our work is that LP-184 sensitivity correlates positively with transcript levels of PTGR1 in the panel of cell lines tested. This supports the hypothesis that PTGR1 is very important for the cytotoxicity of this compound. The third highlight is that LP-184 activity actually turned out to be independent of mutations in the four genes that I mentioned previously, KP1, KRAS, STK11, and TP53. When we compared cell line subgroups with and without mutations in these genes, there was no statistically significant difference in the IC50s across the cell lines. LP-184 retained in vitro activity, even in the presence of deleterious mutations in these genes that would otherwise be associated with chemotherapy resistance in non-small cell lung cancer. The fourth highlight is that LP-184 turned out to be up to 3000 times more potent than commonly prescribed chemotherapy agents such as cisplatin, doxorubicin, gemcitabine, and oxaliplatin.
The fifth highlight was that LP-184 dose ranges that were safe and tolerable were identified in vivo in the mouse model, along with demonstration of anti-tumor efficacy in the xenograft mouse model of non-small cell lung cancer. And the sixth highlight, which is the most clinically relevant, is that elevated PTGR1 cooccurs with mutated KP1 in clinical samples upon retrospect of analysis from clinical databases. Non-small cell lung cancer, especially is one of the very few cancer types in which the response biomarker PTGR1 that underlies LP-184 activation actually correlates very significantly with the molecular drivers of the disease that are often mutated and even undruggable, For example, KP1.
Here we see a clear overlap between the drug activity and the area of unmet need in non-small cell lung cancer. And this also makes sense mechanistically considering the regulatory relationships between components of the KP1 and rf2 PTGR1 access. But we have wild-type KP1 in cancer cells, represses Nrf2 and does not induce PTGR1 above basal levels. However, when KP1 is mutated as seen in up to 20% of lung cancer patients, it does not repress and Nrf2 which is then free to translocate to the nucleus and induce its target genes, including PTGR1. So there you see how KP1 mutation could really drive the regulation of PTGR1, which in turn would sensitize the tumor cell to LP-184.
Now I’d like to talk a bit about what we would like to do next, what’s in progress, and where we would like to see our research being taken.
We find that LP-184 is a promising small molecule with activity in multiple solid tumor types. We continue to identify patient-derived models that corresponds to the clinical and molecular niches that LP-184 is predicted to be effective in. We’d really like to see translation of our cell line based in vitro and in vivo work to a demonstration of efficacy and safety and patient data models, be it ex vivo, 3-D models, fresh tumor biopsies, or xenograft, not only in non-small cell lung cancer, but in other solid tumor types where strong rationale exists to pursue the development of LP-184. Predominantly undruggable non-small cell lung cancer is associated with either KP1 or KRAS mutations that account for 25% of lung cancer patients. And there are very few therapeutic options that are only recently emerging to address these patients. Further, there are also uncharacterized or non-detectable mutations or alterations occurring in up to 35% of non-small cell lung cancer patients.
Now, when we compared overall survival and time to treatment failure with the two subgroups of patients, KP1 mutated and KP1 wildtype receiving the same standard platinum doublet chemotherapy, the KP1 mutant patients perform much worse. Their outcomes are much inferior to the KP1 wildtype patients. Also, KP1 mutant patients are more likely to undergo metastasis. So we’d like to really propose LP-184 as a treatment option in three different areas. One, as an option for KP1 mutated lung cancer. Two, as an alternative to cisplatin in patients that are ineligible to receive platinum-based therapies. And three, as a combination with radiation therapy, as part of the palliative care for late stage cancer patients.
There are other areas also that we envision LP-184 entering the clinic, and those include DNA damage repair, deficient solid tumors, such as pancreatic cancers, having defects in homologous recombination pathway or the nucleotide excision repair pathway. Furthermore, since we have preliminary hints that LP-184 crosses the blood-brain barrier and shows in vivo anti-tumor effects in aggressive brain tumors in mouse models. It also becomes a promising candidate for CNS tumors as well as brain metastasis.
And at the end, I would like to extend acknowledgements to the entire Lantern team of my colleagues, supervisors, and executives, as well as our partners at REPROCELL, without your support this work would not have been possible. Thank you.
Click here to read the full study published by Oncotarget.
YOU MAY ALSO LIKE: More Oncotarget Videos on LabTube
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read—without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues, and other researchers, far and wide.
For media inquiries, please contact media@impactjournals.com.